FULL-LENGTH ARTICLE
Sydney Hopkins* and Nicole L. Mazuroski⁺
School of Health Sciences, Barton College, Wilson, NC, USA
*Student author, ⁺Faculty mentor
CITATION
Hopkins, Sydney; & Mazuroski, Nicole L. (2026). Unraveling neuroblastoma: Causes, treatments, and future perspectives. Barton Journal, 1(1), 96–105. https://bartonjournal.org/vol-1-no-1/2026-cat1-article-no-005
Abstract
Pediatric cancers called neuroblastomas are often seen in children five years and younger. This cancer occurs when the neural crest cells in the adrenal glands, located above the kidneys, do not differentiate properly, resulting in abnormal hormone production and tumor growth. Among neuroblastoma cases, the majority arise as a result of genetic mutations in a few specific genes. Namely, these genes include the anaplastic lymphoma kinase (ALK), paired-like homeobox 2B (PHOX2B), and myelocytomatosis-neuroblastoma (MYCN) genes. In all three, the cancer is formed when such genes are amplified. Currently, there are seven main methods of treatment of the disease, with the primary pharmaceutical option being inhibitors of the ALK gene. In this study, a current ALK inhibitor (Crizotinib) was selected and modified through the alkylation of the piperidine ring located in the drug. This alteration shifted the electron density of the molecule, making it more stable when metabolized, and the lipophilicity of the drug was increased. As a result, ALK, ROS, and MET receptors were blocked through competitive inhibition of the active sites, not allowing ALK 1 and 2 ligands to bind. This pharmaceutical decreases tumor growth and increases apoptosis of cancerous cells through a mechanism change by which the tumor forms. Along with this, the side effects of the new drug showed hypothetical decreases in severity and lower dosage amounts.
Keywords: neuroblastoma, ALK inhibitor, Crizotinib, drug metabolism, ALK gene
Unraveling Neuroblastoma: Causes, Treatments, and Future Perspectives
The neuroblastoma is a form of cancer that arises from genetic mutations. It has been seen mostly in children five years and younger. A special type of cell called the neuroblast is the primary cell type responsible for neuroblastoma. As these cells develop, they do not grow, divide, or differentiate properly, causing tumors to form. In most cases, the cancer is found to be sporadic or hereditary, with different genes mutated that were associated with either form or origin. Cases of neuroblastoma have recently been found to have high incidence rates, with a majority coming from first-world countries, suggesting its significance. The three most common mutations were the ALK, N-MYC, and PHOX2B genes. This study focused on the ALK gene and the mechanism of an ALK inhibitor. In the mechanism, the goal of this project was to create a modified version of the ALK inhibitor chosen, Crizotinib, to enhance the anticancer effects and decrease the onset time of the drug. The ALK gene is responsible for cell growth, and hence, tumor growth as well. Crizotinib was the drug selected for modification due to its being the most common ALK inhibitor prescribed for hereditary neuroblastoma. It also has regions in the drug that are highly susceptible to modification.
The drug was modified through the piperidine ring being alkylated on the electronegative nitrogen atom. A piperidine ring is a carbon-nitrogen cyclic structure that acts as a scaffold in most pharmaceuticals. Through the addition of an alkyl isopropyl group—a three-carbon branched chain—to this ring, the drug became more stable in its electronegativity and electron density. As a result, many positive side effects came about, such as an increase in cell membrane permeability, a decrease in solubility, and a decrease in dosage amount needed. Benefits like these suggest possible modifications that could be made to other anticancer agents or even other medications used in modern medicine.
Process
Neuroblastoma was chosen as the condition of interest due to its significance in childhood cancers. A timeline of significant events, discoveries, and pharmaceuticals developed was created in a previously conducted literature review. After completion, a total history of the condition was observed, and areas of possible improvement were identified. Once a history was taken, genes of interest were researched to discover why they create tumors and how they could possibly be reverted to their original state. The way the genes were reverted was through pharmaceutical use.
A drug was selected for modification with the criteria that it is a) prevalent for neuroblastoma treatment, b) is capable of being modified, and c) has dangerous side effects that could be diminished. Crizotinib was the perfect option as such. This drug acts as an inhibitor of one of the major genes mutated in neuroblastoma (ALK) to create an anticancer effect (Crizotinib, n.d.). When the drug was observed, a few areas were noted as being good starting points for modification. Such areas included the piperidine ring, multiple halogens, and a pyrazole ring. The pyrazole ring is another cyclic carbon-nitrogen ring in the compound. Next, a list of possible modifications was posed, which resulted in the isopropyl alkylation of the piperidine ring being chosen. The mechanism for how this could be done was established, and possible benefits were further analyzed.
Project Selection
Across the globe, neuroblastoma is the leading form of cancer in infants and children (National Cancer Institute, 2024). Such cancer is caused by hereditary or sporadic factors. This idea suggested there were possible ways to prevent hereditary forms from occurring if the genetic mutations could either be reversed or underexpressed. The way this is done is through pharmaceutical drug modification. In such pharmaceuticals, drugs often present negative side effects, but can be altered using chemistry. Drug modification has been a driving force in healthcare, thereby suggesting that theoretical mechanisms have become more prevalent in pharmacy.
Background and Prior Work
Neuroblastoma, as a form of cancer, was first discovered by a German physician named Rudolf Virchow in 1864 upon discovering abdominal gliomas (Mandal, 2019). Such tumors developed in the adrenal glands and sympathetic nervous system, showing a linkage to neural cells. Specifically, the cells that cause the tumor are identified as neuroblasts, which are a cell type located in the adrenal medulla of the adrenal gland. Such cells are immature in the sense that they are undifferentiated (ACCO, 2022). When cells mature, they transform into cells with a select few responsibilities and are generally located in specific regions of the body. Specifically in neuroblastoma, the cells have not differentiated, meaning they are still stem cells, but have no specific function in the adrenal glands. Due to this, the cancerous cells often secrete higher levels of hormones, like catecholamines, which can be used diagnostically for adrenal tumors (Mandal, 2019).
Since neuroblastoma was first discovered, the incidence rates globally have increased dramatically among children. According to the Cleveland Clinic, this linear increase from 1990 to 1999 and again in 2005-2018 is likely due to hereditary factors (2025). Neuroblastoma has shown no evidence of being carcinogenic, meaning there has not been a specific chemical/substance shown to cause gene mutations resulting in cancer (Cleveland Clinic, 2025). For a cancer to be carcinogenic, it means the cancer is caused by a certain substance or chemical that a person might be exposed to. A common example is nicotine being a carcinogen for lung cancer. The incidence percentage rate from 1990 to 2021 (Figure 1) depicts the trend of cases being diagnosed internationally (Nong et al., 2024). One note of significance was the sharp decline from 2018 onward. A decline like this was likely due to the COVID-19 pandemic and a shift in the focus among healthcare providers to treating patients afflicted with the virus (Nong et al., 2024). It could also have been as a result of more treatments being released around that time period.
Figure 1
Incident Percent Rate Change of Childhood Neuroblastoma 1990-2021
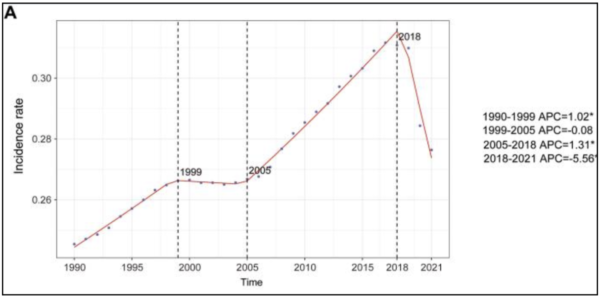
Note. Annual percent change (APC) and trends in global childhood neuroblastoma incidence from 1990 to 2021.
A global image showing the incidence rate of neuroblastoma per country (Figure 2) illustrated the prevalence of neuroblastoma worldwide (Nong et al., 2024). While there are some countries with a low rate of diagnosis, most other countries, especially first-world countries, showed the highest values. This trend likely occurred because of the higher quality of healthcare. A majority of third-world countries do not have the equipment or personnel to diagnose such conditions, potentially contributing to lower rates. There has not been substantial evidence suggesting this, so it was a theory established by the researchers. Even with this in mind, both demographics aided in providing evidence of neuroblastoma having great significance, yet few solutions in the medical field.
Figure 2
Global Demographic of Childhood Neuroblastoma Incidence
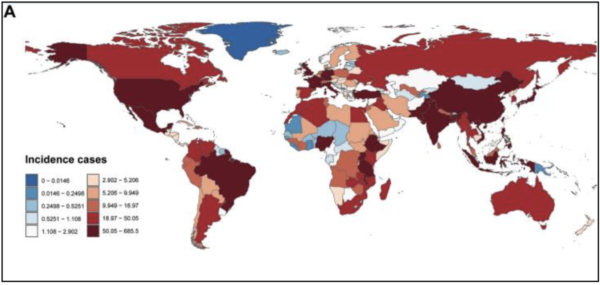
Note. Incidence of childhood neuroblastoma across 204 countries and territories. Number of incidents.
In 1896, X-rays were used as an imaging technology to identify internal tumors, specifically, for neuroblastoma (National Cancer Institute, 2024). X-ray machines produce electromagnetic radiation waves that travel through the patient and hit a wall behind the patient, showing tissue density. Tissues that have a higher density, such as bones, absorb more of the rays, which would produce a lighter color. Lighter tissues, such as skin, do not absorb many rays and would produce a darker color on the scan created.
The N-MYC gene was identified as one of the primary target genes of neuroblastoma in the early 1980s, with the first human antibody treatment for neuroblastoma being discovered (Furman, 2021). This gene region is responsible for cell growth, division, and differentiation through its acting as a transcription factor for DNA and controlling other gene expression. Such a gene as this is labelled as proto-oncogenic, which means the gene could cause cancer (National Cancer Institute, n.d.). When a mutation occurred, the gene was overexpressed, and tumors were produced, causing cells to become oncogenic. With the N-MYC gene, overexpression led to a worse prognosis for the patient after diagnosis. Along with the overexpression, it was commonplace to see a resistance to ALK inhibitors in such cases (Bachetti et al., 2010).
Other genes for neuroblastoma (ALK and PHOX2B) were later discovered in the human genome mapping project in 2008 (Barr & Applebaum, 2018). Much like the N-MYC gene, the ALK gene is responsible for producing the protein ALK receptor tyrosine kinase (ALKRTK), regulating cell growth (Wulf et al., 2021). Most often seen are point mutations of the ALK gene, resulting in overexpression of the gene in sporadic neuroblastomas (Holla et al., 2017). A point mutation is one where one nucleotide is changed, affecting the reading of the amino acid sequence produced. A disruption in this reading causes small ligands called augmentor α and β to over-attach, thus causing overexpression (Shreenivas et al., 2023). When the ALK gene is mutated, tumor growth is the primary effect created (Shreenivas et al., 2023). It is also important to note that ALK mutations often result from hereditary mutations.
The third gene of significance, which is similar to the ALK gene, is the PHOX2B gene. While being attributed to neuroblastoma, the PHOX2B gene focuses on division and differentiation of the neuroblasts (Hereditary Neuroblastoma, n.d.). Neuroblasts are located in the neural crest of the adrenal gland, which is a region that specifically houses these stem cells in fetuses and infants (MFMER, 2025). When mutated, either the amino acid sequence is altered, or several nucleotides are switched, which results in incorrect amino acids being inserted into the gene (U.S. National Library of Medicine, 2019). When this occurs, the neuroblasts improperly differentiate, thus causing tumors to form. It was discovered that each of these three genes is a prevalent cause of the initial tumor formation when mutated.
Once such genes were identified, possible treatments began to be proposed. In 2020, the first monoclonal antibody treatment for neuroblastoma was developed and approved for clinical use. Coined as Danyelza®, the drug targets antigens present on the tumor cells and produces antibodies naturally by the body. The specific antigen is a disialoganglioside antigen, meaning there are acidic residues on the cancer cell affecting its growth and adhesion to other cells.
Methods
Research was conducted to begin as a background of neuroblastoma needed to be established. Once complete, the causes of neuroblastoma were identified. Initially, it was thought there could be a possible carcinogen associated with the condition. However, this was quickly dismissed as all research conducted did not point to a carcinogen causing mutation of the genes responsible for neuroblastoma. After it was determined that neuroblastoma is only caused by genetic mutations, this path was taken.
Drug Modification. Through the identification of the genes associated with neuroblastoma, the treatment options used were then evaluated. It was observed that pharmaceuticals were most often selected as the course of treatment. Using this knowledge, the most common ALK inhibitor prescribed was called Crizotinib and was selected for modification. This drug suggested multiple opportunities for adjustment to reduce harmful side effects and increase effectiveness.
Upon further inspection of the drug, the piperidine ring was suggested as the best area of modification. The reason is due to it having an electronegative nitrogen atom and its structure causing a scaffolding effect in drugs. The orientation of the ring suggests a possible addition of an alkyl group onto the nitrogen. An alkyl group is one that is a hydrocarbon chain, and increases lipophilicity and decreases solubility. Knowing this, a mechanism was created and tested for possible side effects that would occur in the patient. The mechanism was carried out using a base of potassium carbonate (K2CO3). Potassium carbonate acted as a weak base and was able to deprotonate the nitrogen atom. Deprotonation is the act of removing a proton from an atom/molecule. Such results from the mechanism were compared to the original effects of Crizotinib to establish whether the mechanism worked correctly and if the results aligned with the desired effects.
Outcomes
The new mechanism and altered form of Crizotinib were tested for impacts on stability and possible clinical effects within the patient. It was discovered that the mechanism itself is indeed possible with a near one hundred percent yield of the new drug created. These results are likely due to the use of a weak base that does not cleave the ring apart into different components. Additionally, it can be reasoned that the piperidine acting as a scaffold is more reactive than other elements in the drug under the conditions created for alkylation. Due to there being only one hydrogen present on the more electronegative—a chemical property of an element’s affinity for lone electrons—nitrogen, this provided the best area for substitution to occur.
There has been no mechanism proposed for this drug to be modified in the manner executed, suggesting alkylation might be more difficult to execute than hypothesized. In other alkylated drugs, results showed that a majority of common chemotherapy drugs were alkylated to increase the anticancer therapy. Alkylation causes DNA (deoxyribonucleic acid) to be disrupted in structure and function, ultimately leading to cell death (Drugs.com, 2025). Since this has been done to other drugs, it was reasonable to theorize that the anticancer effects would be increased as a result of the new mechanism and alkylation. With the goal in mind of enhancing a drug already on the market, the alkylation of this drug fulfilled the goal set.
Regarding other significant characteristics of interest, many of them were altered to give the desired effects. Alkylation of the piperidine ring increased the lipophilicity—a substance’s tendency to cross cell membranes that have a phospholipid bilayer—of the drug when ingested. The phospholipid bilayer contains a hydrophobic tail and a hydrophilic head, which allows the membrane of cells to be selectively permeable to certain molecules (Alberts, 1970). The presence of more carbon and hydrogen atoms caused more polar qualities to come about, which include high lipophilicity and low solubility. When compared with previous literature, a higher lipophilicity is likely to cause the drug to pass through the cell membrane more easily, thus decreasing the onset time of the drug.
Additionally, the solubility—how quickly the drug was metabolized—was decreased. When a substance enters the bloodstream, it begins to be broken down until it can no longer be broken down, and at that point, it is ready for elimination. Because the goal was to enhance the drug’s already created effects, a decreased solubility indicated the drug was not broken down immediately after entering the bloodstream. Hereby, the anticancer effects were able to be prolonged, which also allowed dosage amounts to be lower. For the patient, this meant that they would not have to receive treatment either as much or in as many doses as the standard.
Interestingly enough, there were not many obstacles that needed to be tackled when creating the drug mechanism. There was only one significant factor, and that was the selection of the drug for modification. The original idea was to use a different drug that inhibited the N-MYC gene instead of the ALK gene, but the common drugs used for that purpose were not modifiable (Whitfield & Soucek, 2021). Such an issue is not severe, though, as this was the first step of analyzing the drug to create the metabolism. More research was conducted until it was found that ALK inhibitors, specifically Crizotinib, were a good match for drug modification.
Pharmaceuticals can be modified in many ways, and these are dependent on the structure and functional groups within the pharmacophore—the active portion of a drug. Crizotinib was a good drug to select, as many areas of the drug are subject to change, like electronegative atoms and nitrogenous rings. The use of a weak base and a haloalkane—organic compound with a hydrocarbon chain containing some halogen atom acting as the initiator for the reaction—served to alkylate the nitrogen inside the piperidine ring. Therefore, the new drug enhanced certain anticancer effects already created. Such effects included lipophilicity, solubility, and dosage quantity for patients. It was through the alkylation that the lipophilicity was increased, which allowed the drug to pass more quickly through the cell membrane and begin anticancer effects sooner. The lower solubility caused the drug to not be metabolized by hepatocytes—cells located in the liver—as quickly, prolonging the anticancer effects (Pfizer, n.d.). In doing so, the dosage amount prescribed could be lowered, which could make life slightly easier for the patient. Drug modification is an area of pharmaceuticals still being discovered, and through mechanisms such as the one created, drugs are being improved, and more patients are being saved.
Future Directions and Applications
Future implications include carrying out the new mechanism in a live experiment in the laboratory to see if the yield is correct and whether the product created is the desired hypothesized drug. The product would be tested using an FT-IR spectrometer (Fourier- Transform infrared spectrometer) instrument that is able to analyze a sample and determine the structure of the analyte very closely. An experiment such as this would need to be completed at an institution or facility with proper equipment to do so. In the field of drug discovery and modification, simulations are barely being used to test for side effects. One of the innovations with AI is to test mechanisms before drugs undergo living trials. It allows for the testing in humans without potentially harming humans or animals. Within the field of pharmacy, anyone who graduates with a PharmD degree is able to become an industrial pharmacist and work on projects exactly like this one, except with the real compounds and machines. During rotations in pharmacy school, a majority of candidates will take rotations in such places, so even having a theoretical project such as this is extremely beneficial.
Conclusion
Neuroblastoma is a form of cancer prevalent in young children. It arises from improperly differentiated neuroblasts and is often caused by genetic mutations. The most common genes altered are the ALK, N-MYC, and PHOX2B genes. All three of these genes are overexpressed in neuroblastoma, causing abnormal cell growth, division, and differentiation. For the ALK gene, drugs identified as ALK inhibitors are used to reduce the overexpression caused by the mutation. The most common ALK inhibitor, known as Crizotinib, can be modified using different reagents to alter the functional groups of the pharmacophore, which changes certain side effects. With the objective in mind of adjusting this pharmaceutical for neuroblastoma to obtain desired results physiologically, a new mechanism was created to slightly alter the drug. The region altered was the piperidine ring, which was alkylated with an isopropyl group. This alkylation resulted in a higher lipophilicity, lower solubility, and lower drug dosage for patients. Drug modification, as witnessed here, is a field of pharmacy being thoroughly explored to enhance drugs already on the market by improving side effects and improving the quality of life for patients undergoing all forms of treatment. The field of medicine would benefit greatly from alterations like this, and many more conditions could become treatable after such alterations.
References
ACCO. (2022, March 10). Childhood neuroblastoma cancer: Causes, risk factors, and prevention. American Childhood Cancer Organization. https://www.acco.org/ blog/childhood-neuroblastoma-cancer-causes-risk-factors-and-prevention/
Alberts, B. (1970, January 1). The lipid bilayer. Molecular Biology of the Cell (4th ed.). https://www.ncbi.nlm.nih.gov/books/NBK26871/
Bachetti, Tiziana; Di Paolo, Daniela; Di Lascio, Simona; Mirisola, Valentina; Brignole, Chiara; Bellotti, Marta; Caffa, Irene; Ferraris, Chiara; Fiore, Michele; Fornasari, Diego; Chiarle, Roberto; Borghini, Silvia; Pfeffer, Ulrich; Ponzoni, Mirco; Ceccherini, Isabella; & Perri, Patrizia. (2010). Phox2b-mediated regulation of ALK expression: In vitro identification of a functional relationship between two genes involved in neuroblastoma. PLoS ONE, 5(10). https://doi.org/10.1371/journal.pone.0013108
Barr, Erin K.; & Applebaum, Mark A. (2018). Genetic predisposition to neuroblastoma. Children, 5(9), 119. https://doi.org/10.3390/children5090119
Cleveland Clinic. (2025, March 19). What you need to know about carcinogens. Cleveland Clinic. https://my.clevelandclinic.org/health/articles/25081-carcinogens
DrugBank. (n.d.) Crizotinib: Uses, interactions, mechanism of action | drugbank online. DrugBank. Retrieved April 05, 2025, from https://go.drugbank.com/drugs/DB08865
Drugs.com. (2025, April 13). List of alkylating agents. Drugs.com. https://www.drugs.com/ drug-class/alkylating-agents.html
Furman, Wayne L. (2021). Monoclonal antibody therapies for high-risk neuroblastoma. Biologics: Targets and Therapy, 15, 205–219. https://doi.org/10.2147/btt.s267278
Holla, Vijaykumar R.; Elamin, Yasir Y.; Bailey, Ann Marie; Johnson, Amber M.; Litzenburger, Beate C.; Khotskaya, Yekaterina B.; Sanchez, Nora S.; Zeng, Jia; Shufean, Abu; Shaw, Kenna R.; Mendelsohn, John; Mills, Gordon B.; Meric-Bernstam, Funda; & Simon, George R. (2017). ALK: A tyrosine kinase target for cancer therapy. Molecular Case Studies, 3(1). https://doi.org/10.1101/mcs.a001115
Mandal, Ananya. (2019, February 27). Neuroblastoma history. News Medical. https://www.news- medical.net/health/Neuroblastoma-History.aspx
MFMER. (2025, February 25). Neuroblastoma. Mayo Foundation for Medical Education and Research. https://www.mayoclinic.org/diseases-conditions/neuroblastoma/ symptoms-causes/syc-2035101
National Cancer Institute. (n.d.). NCI Dictionary of Cancer Terms. National Cancer Institute. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/proto-oncogene
National Cancer Institute. (2024, October). Neuroblastoma treatment. National Cancer Institute. https://www.cancer.gov/types/neuroblastoma/patient/neuroblastoma-treatment-pdq
Nong, Jusen; Su, Cheng; Li, Changhua; Wang, Congjun; Li, Wei; Li, Yong; Chen, Peng; Li, Yanqiang; Li, Zihao; She, Xinjin; Yuan, Zuxin; Liu, Sentian; Chen, Chao; Liao, Qian; Luo, Yige; & Shi, Bo. (2024). Global, regional, and national epidemiology of childhood neuroblastoma (1990–2021): A statistical analysis of incidence, mortality, and DALYs. eClinicalMedicine, 79, 102964. https://doi.org/10.1016/ j.eclinm.2024.102964
Pfizer. (n.d.). Xalkori® (crizotinib) Clinical Pharmacology: Pfizer medical information – US. Pfizer. Retrieved February 27, 2025, from https://www.pfizermedicalinformation.com/ xalkori/clinical-pharmacology
Shreenivas, Aditya; Janku, Filip; Gouda, Mohamed A.; Chen, Hui-Zi; George, Ben; Kato, Shumei; & Kurzrock, Razelle. (2023). ALK fusions in the pan-cancer setting: Another tumor-agnostic target? Npj Precision Oncology, 7(1). https://doi.org/10.1038/s41698-023-00449-x
St. Jude Children’s Hospital (n.d.) Hereditary neuroblastoma. St. Jude Children’s Hospital. Retrieved December 29, 2024, from https://www.stjude.org/care-treatment/treatment/ genetic-syndromes/hereditary-neuroblastoma.html
U.S. National Library of Medicine. (2019, September 1). Phox2b gene: MedlinePlus Genetics. MedlinePlus. https://medlineplus.gov/genetics/gene/phox2b/
Whitfield, Jonathan R., & Soucek, Laura. (2021). The long journey to bring a MYC inhibitor to the clinic. Journal of Cell Biology, 220(8). https://doi.org/10.1083/jcb.202103090
Wulf, Anna M.; Moreno, Marcela M.; Paka, Chloé; Rampasekova, Alexandra; & Liu, Karen J. (2021). Defining pathological activities of ALK in neuroblastoma, a neural crest-derived cancer. International Journal of Molecular Sciences, 22(21), 11718. https://doi.org/10.3390/ ijms222111718

