SHORT ARTICLE
Savannah Olson* and Nicole L. Mazuroski⁺
School of Health Sciences, Barton College, Wilson, NC, USA
*Student author, ⁺Faculty mentor
CITATION
Olson, Savannah, & Mazuroski, Nicole L. (2026). Understanding radiographic chemistry. Barton Journal, 1 (1), 135–141. https://bartonjournal.org/vol-1-no-1/2026-cat2-article-no-010
Abstract
This study researched radiographic compounds with the intention of gaining a better understanding of the chemical structure and mechanism of action of radiographic contrast agents. Through a series of experiments and research methods, conclusions have been drawn about the properties of contrast agents and their effects on the body and medical images. Before working with chemical agents, ample research was done on the composition and chemistry of radiographic contrast agents. Laboratory experiments were then produced and executed using smartphone colorimetry as a tool to receive RGB values for various solutions. These labs were created with the intention of using compounds that make up radiographic contrast agents to observe reactions in a controlled, less invasive environment. Experimental results align with the research conducted, indicating the solubility, redox behavior, and chemical properties of contrast agents. The findings can be used to deepen knowledge of contrast agents and, in turn, have an impact on medical technology; the eyes of medicine.
Introduction
This study will analyze the role of contrast agents in radiographic chemistry by examining the mechanisms of action and chemical stability. Conducted as a semester-long project, it involved testing and analyzing contrast agents in the lab. The work consisted of a combination of research methods and a physical lab component over the course of twelve weeks. Iodinated contrast media contain iodine and are used in X-ray techniques, including CT (Murphy et al., 2026). The primary administration of iodine-based contrast agents is through an IV, though they can also be administered differently depending on the test type or patient needs. Due to their high atomic number, iodinated contrast agents produce adequate contrast of tissues in the body, increasing image quality. Iohexol is one contrast agent that is used in computerized tomography. Another major type of contrast agent used in medical imaging is barium-based contrast agents. Typically, these agents are used for images of the lining of the gastrointestinal tract but can be dangerous if there are perforations in the bowel. Barium-based agents are administered rectally or orally, as opposed to intravenously like iodine-based contrast agents (Noah Chemicals, 2024).
Additionally, this study primarily focuses on the mechanism of action and chemical stability of iodine-based contrast agents. Each type of contrast agent has its own chemical composition that makes it more efficient for different medical imaging types. Knowing the chemical stability of different agents helps determine which agents to use for each test and explains why they are used for images in different body regions. Iodinated contrast media are either high or low osmolality, which refers to the rate at which water diffuses. High osmolality contrasts are specifically dangerous near the respiratory system due to a high risk of pulmonary edema (Zeligman, 2010). Side effects like this are crucial for healthcare providers to be aware of to prevent patients from being harmed or dying. These contrasts are safe for medical imaging that does not require administration by swallowing or through a tube (Zeligman, 2010).
Iodinated contrast media are also commonly used in the field of radiology due to the excellent medical images that can be produced by iodine’s ability to absorb X-rays and make internal bodily structures visible (Yang et al., 2018). Iodine’s high atomic number allows photons to be absorbed, and structures like blood vessels appear bright white on the images. Researchers are constantly working to find ways to adapt contrast agents to create the highest visibility in medical images to better diagnose diseased areas of the body. Many Magnetic Resonance Images (MRIs) use contrast agents to aid in this process. MRIs are used as a diagnostic tool to reveal inflammation, tumors, and tissue abnormalities (Lv et al., 2023). It is important for contrast agents used for MRIs to have the ability to polarize water molecules and yield a high-contrast, high-quality image that can be used to diagnose diseases. Due to the composition of the human brain, different contrast agents may be needed, as contrast is lower in the brain than in other areas of the body (Lv et al., 2023). Body parts and mechanisms of action should be considered when determining which contrast agents to use for different diagnostic radiographic procedures.
Methods & Materials
The first procedure completed was a smartphone colorimetry experiment. The Redox of Iodine Using Apple Juice and Smartphone Colorimetry lab was created and produced to showcase the change iodinated compounds encounter over time when a Vitamin C source is added. The purpose of this study is to gain a better understanding of the core chemistry behind contrast agents. Materials needed included 0.05M iodine, starch solution, apple juice, water, small beakers, pipettes, white paper, and an iPhone camera. To prepare the starch indicator, 1 teaspoon of cornstarch was mixed with 20 mL of warm water in a small beaker and stirred until cloudy. The iodine-starch complex was then created by adding 5 mL of iodine and 3 mL of starch solution. This mixture was stirred until it became a deep blue hue. Next, 3 mL of apple juice was added as the reducing agent. The solution was swirled and observed. The cup was placed onto a white sheet of paper as the background, and smartphone colorimetry measurements were taken every two minutes using a color-analyzer app. Each time a new picture was taken, the RGB values were recorded. Iodine is a stable element, but it can exist in various forms. When Vitamin C is added, iodine is reduced to iodide. Fortunately, Vitamin C levels in the body are not high enough to reduce iodine in iodine-based contrast agents on their own, so there is not an effect on imaging quality based on normal values. Vitamin C has been tested and is believed to be a radioprotector. Studies conducted on mice introduced ascorbic acid to the organism, where it acted as a radioprotector (Narra et al., 1993). While there is still ample research to be done, this could be a monumental finding in medicine if Vitamin C can be used to protect humans from radiation in the future.
A second experiment completed during this study was Partitioning Water vs. Oil to determine how hydrophilic iodine is. Iodine-based contrast agents are designed to be hydrophilic to allow for safe dissolution into the bloodstream and removal via the kidneys. Iodinated contrast agents are commonly composed of tri-iodinated benzene rings (Murphy et al., 2026). This structure allows the contrast agents to be highly water-soluble and safe for administration in diagnostic radiographic imaging. The protocol demonstrates solubility and lipophilicity, explaining why many contrast agents are formulated as water-soluble. Materials used included iodine solution, corn oil, test tubes, pipettes, starch solution, and smartphone colorimetry. One vial was labeled Before and one was labeled After. In the Before vial, distilled water and iodine solution were combined, and a baseline photograph was taken. In the After vial, distilled water, iodine solution, and vegetable oil were combined, then shaken to allow partitioning. After sitting for 10 minutes, the layers separated. A sample from the aqueous layer was removed and analyzed using the same colorimetry method. This procedure demonstrated lipophilicity, hydrophilic properties, and osmolality of iodinated contrast agents. Some common contrast agents used for CT scans, categorized as low osmolality and highly water-soluble, include iohexol, iopamidol, iopromide, ioversol, and ioxilan (Murphy et al., 2026).
Results & Discussion
In the first experiment, the solution was observed over several minutes, and the RGB values provided by the smartphone colorimetry app were recorded. Values of importance in this lab were the Blue values. Iodine was converted to iodide in this reaction, which does not bind to starch and led to the color fading. As minutes passed, the blue concentration became lower, which was expected with Vitamin C added into the iodine-starch solution (Figure 1). The blue value fell from 211.0 to 182.9 to 102.0 to 98.3, 93.1, 86.8, 80.8, 54.9, 43.4 to 43.4. Over 18 minutes, the blue value fell by 167.6. This color change is not visible to the human eye, which is why the smartphone colorimetry app is needed to detect numerical values. Vitamin C reduces iodine to iodide and dilutes it, which reduces the blue hue of the solution. As time passed, the iodine became more diluted and less concentrated. Although the addition of Vitamin C to iodine does not directly affect radiographic image quality, it does change the chemical stability of iodine. If anything alters the state of iodinated contrast agents, it could affect how they react in the body and how well they contrast bodily structures in medical images. The blue RGB value is measured since the yellowish/brown color of iodine absorbs light in the blue wavelength. Vitamin C can also lessen the negative effects of iodinated radiation. While iodinated contrast agents themselves do not expose people to radiation, imaging techniques such as X-rays do (Mustafa, 2024). Understanding iodine chemistry is important, as it can be reduced with Vitamin C to help prevent radiation damage. Healthcare professionals can use this knowledge to help patients and potentially reverse radiation exposure if needed.
Figure 1
Blue Value Changes Over Time

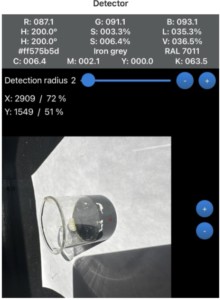
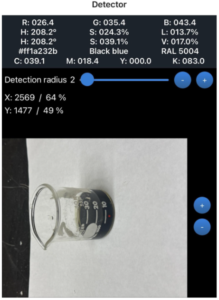
Note. Shown at initial stage, 8 minutes, and 16 minutes.
Results of the second portion of the physical lab component are shown below. These results demonstrate that most iodine remained in the aqueous phase, relating to radiographic contrast agents that are designed to be hydrophilic and remain in the blood. Iodine is water-soluble and dissolves into the bloodstream, moving through it before being excreted by the kidneys. This property is a key component of iodinated contrast agents. Oil-based contrast agents, however, are used in radiology for high visibility and specialized imaging. Their chemical makeup differs from iodine-based agents. Being hydrophobic, they do not dissolve in water, which is the opposite behavior of iodinated contrast media. This lab demonstrates how oil-based contrast agents form layers when distributed among substances. Such layering accurately mimics how these agents behave in the body. Oil-based agents are commonly used to capture images of the fallopian tubes and uterus, as partitioning produces high visibility and allows them to remain in the system longer than other types of contrast agents (Morita et al., 2025). They may also be injected into ducts to detect tumors and are often preferred because of their extended retention time. Additionally, their chemical structure contributes to high contrast and viscosity, leading to clearer images and aiding diagnosis. Some concerns exist with the use of oil-based contrast agents. In one study involving a pregnant patient who underwent an abdominal radiograph, complications were observed. Because these agents remain in the body longer, they may be identified as foreign objects (Morita et al., 2025). Although the complications in that case were not harmful, the response raises concern about how the body may react.
Figure 2
Results Showing Iodine Remained in the Aqueous Phase
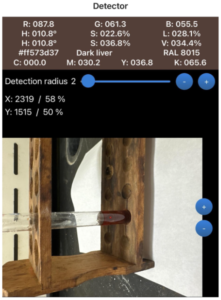
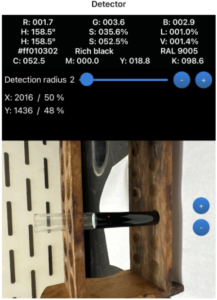

Note. Shown are the Before vial, After vial, and label removed.
Conclusion
In the Vitamin C colorimetry experiment, iodine and starch created the blue complex, and apple juice contained ascorbic acid, the reducing agent in the reaction. Iodine converts to iodide, which does not bind to starch, causing the color to fade. The solution began dark blue and progressively faded. Similar to observations in this lab, radiographic contrast agents contain iodine and change chemically in predictable time courses. Using iodine in a controlled environment allowed for the mimicking of processes of radiographic contrast agents. The corn-oil diffusion lab models how radiographic contrast agents behave in the body. It demonstrated how a hydrophobic, fat-soluble substance navigates an aqueous environment. Corn oil is hydrophobic and separates rather than dissolving in water. A similar reaction occurs in oil-based contrast agents. Immiscible liquids divide into layers, which is also what happens to contrast agents that remain in different tissues.
The Vitamin C smartphone colorimetry lab measured the concentration of Vitamin C and yielded a redox reaction. A smartphone colorimetry iPhone application was used to gain qualitative and numerical data for analysis. This tool was very useful in determining if color had faded, as chemical changes are not always apparent to the naked eye. RGB values provide ample information and allow observation of the rate of fading. The reaction was completed to mimic measuring contrast dosage and absorption. The corn oil diffusion lab measured solubility and physical distribution, showing the difference between hydrophilic and hydrophobic reactions. Results can be observed visually through the separation of oil from the aqueous solution. Layering observed in this lab is comparable to layering that occurs in radiographic contrast agents within human tissues.
Research on radiographic contrast agents, along with laboratory procedures, was carried out throughout the semester with the goal of understanding chemical structure and how components influence function. It is important to note margins of error when conducting laboratory work and drawing conclusions. During earlier stages of the semester, the same experiments were performed, but results were not as expected. Encountering obstacles and making mistakes is common while learning, and documenting factors that may influence results is essential. In the first trials, user error occurred while working with the smartphone colorimetry app. Data presented in this report reflects results from the final experiments.
References
Lv, Jie; Roy, Shubham; Xie, Miao; Yang, Xiulan; & Guo, Bing. (2023). Contrast Agents of Magnetic Resonance Imaging and Future Perspective. Nanomaterials (Basel, Switzerland), 13(13), 2003. https://doi.org/10.3390/nano13132003
Morita, Akari; Kakinuma, Toshiyuki; Segawa, Arimi; Harada, Satoshi; Takae, Seido; Tamura, Midori; & Suzuki, Nao. (2025). Prolonged retention of oil-based iodinated contrast medium observed on plain abdominal radiograph after cesarean section: A case report. World Journal of Clinical Cases, 13(29), 110454. https://doi.org/10.12998/wjcc.v13.i29.110454
Murphy, Andrew; Wilczek, Mateusz; Campos, Arlene; Sharma, Rohit; Bell, Daniel J.; Scappatura, Giuseppe; Hutson, Russell, Bickle, Ian; Jones, Jeremy; Johnston, Aimee; Knipe, Henry; Deng, Francis; Hacking, Craig; and Weerakkody, Yuranga. (2026). Iodinated contrast media. Reference article, Radiopaedia. https://doi.org/10.53347/rID-48582
Mustafa, Dunya Ali; Abdul Jabbar, Hind Moafak; Joudha, Luma Naji; & Radhi, Muhammed Mizher. (2024). Evaluation study: the effect of using ascorbic acid with and without contrast agent on patient blood samples exposed to different x-ray by cyclic voltammetry. Al-Nisour Journal for Medical Sciences, 6(2), Article 2. https://doi.org/10.70492/2664-0554.1001
Narra, V. R.; Howell, R. W.; Sastry, K. S.; & Rao, D. V. (1993). Vitamin C as a radioprotector against iodine-131 in vivo. Journal of nuclear medicine : official publication, Society of Nuclear Medicine, 34(4), 637–640. https://pubmed.ncbi.nlm.nih.gov/8455081/
Noah Chemicals. (2024, August 14). The essential role of barium sulfate in medical imaging. Noah Chemicals. https://www.noahchemicals.com/the-essential-role-of-barium-sulfate-in-medical-imaging/
Yang, Jai-Sing; Peng, Yan-Ru; Tsai, Shih-Chang; Tyan, Yeu-Sheng; Lu, Chi-Cheng; Chiu, Hong-Yi; Chiu, Yu-Jen; Kuo, Sheng-Chu; Tsai, Yuh-Feng; Lin, Ping-Chin; & Tsai, Fuu-Jen. (2018). The molecular mechanism of contrast-induced nephropathy (CIN) and its link to in vitro studies on iodinated contrast media (CM). BioMedicine, 8(1), 1. https://doi.org/10.1051/bmdcn/2018080101
Zeligman, E. Bernard. (2010). Chapter 67 – Radiography and radiographic-flouroscopic contrast examinations. GI/Liver Secrets, 23(1), 477–488. https://doi.org/10.1016/B978-0-323-06397-5.00067-8

